 |
A case of a man with hemoptysis and weight loss |
- History of Present Illness
- A man in his 70s presented with cough and weight loss for a month with new onset-hemoptysis. He denies chest pain, dyspnea, fevers, or chills.
- Past Medical History
- The patient has a history of non-muscle-invasive bladder cancer (NMIBC) treated with a transurethral bladder tumor resection (TURBT) about 6 years prior to admission, followed by intravesical BCG installation a few years prior to admission, remote squamous cell carcinoma of the neck treated with radiation, hypertension, and a recently diagnosed proximal aortic arch pseudoaneurysm for which he was undergoing evaluation for thoracic endovascular aortic repair (TEVAR) with a graft.
- Medications
- None.
- Epidemiological History
- The patient was born and raised in the southwestern United States, and currently lives in the rural Pacific northwest. He had traveled in Asia. He enjoys gardening and fishing. He has no history of tobacco, alcohol, or injection drug use. He lives with his wife and dogs.
- Physical Examination
- The patient appeared well. He had a temperature of 99.1 F/ 37.3 C, blood pressure of 189/95 mmHg, and heart rate of 76 beats/minute. On auscultation, he was noted to have diminished breath sounds in the left upper lung field and intermittent cough. The rest of the physical exam was otherwise normal.
- Studies
- The routine laboratory studies, including complete blood count and metabolic profile were within normal limits. Computed tomography (CT) scan of the chest showed a descending aortic arch pseudoaneurysm measuring 1.2 x 1.0 cm. He was also noted to have a left upper lobe lung infiltrate measuring 4.4 x 2.9 cm, adjacent to the pseudoaneurysm and micronodules in the left upper and lower lobes. No microbiologic studies were done at that time.
- Clinical Course Prior to Diagnosis
Due to the suspicion that a fistula between the pseudoaneurysm and bronchus was causing his hemoptysis, the patient underwent an urgent thoracic endovascular aortic repair (TEVAR) with a graft, and no fistula was found. Hemoptysis resolved soon after this procedure.
A follow-up CT two months after the TEVAR showed persistence of the left upper lobe infiltrate, now resembling a mass, demonstrating this was not due to the aortic pseudoaneurysm. The patient underwent work-up for a suspected malignancy. Cystoscopy was negative for malignancy. Fluorodeoxyglucose positron emission tomography (FDG PET) scan showed increased metabolic activity within the left upper lobe, left first rib at the costovertebral junction, left transverse process of the T1 vertebrae, and T2 to T4 thoracic vertebral bodies (Figures 1 and 2). A magnetic resonance imaging (MRI) of the brain showed multiple punctate foci of enhancement in the supratentorial and infratentorial areas of the brain. He also underwent infectious workup at this time including serologies for a variety of endemic fungal, atypical/zoonotic infections (including Brucella spp., Coxiella spp., Toxoplasma gondii), bacterial blood cultures, cryptococcal antigen testing, and fungal blood cultures, all of which were negative.
Bronchoscopy with fine-needle aspiration (FNA) and bronchoalveolar lavage (BAL) was performed to evaluate the mass. Microscopic examination of the biopsies showed necrotizing mucosa and caseating granulomatous inflammation with no malignant cells. Bacterial cultures from the BAL samples grew Streptococcus salivarius and alpha-hemolytic Streptococci. No acid-fast bacilli (AFB) or fungal organism was identified.
CT-guided biopsy of the vertebral body was performed, and it showed non-necrotizing granulomas with epithelioid histiocytes without evidence of malignancy. Bacterial, fungal, and AFB cultures and smears of the bone biopsy were negative.
After an exhaustive workup, he had a follow-up CT scan five months after TEVAR that showed an enlarging left upper lobe mass measuring about 3.2 x 5.2 x 8.1 cm, increased in size craniocaudally to 8.1 cm from 4.6 cm on prior examination (Figure 3).
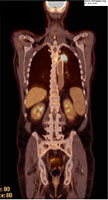 - Figure 1: FDG PET scan; Increased metabolic activity in posterior medial left upper lobe mass; extending into L hilar region.

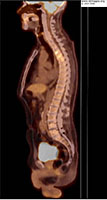 - Figure 2: FDG PET scan; Multiple foci of metabolic activity involving the T1-T4 four vertebral bodies.

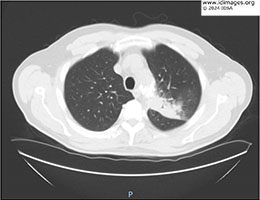 - Figure 3: CT scan of the chest without contrast; Left upper lobe infiltration closely related to aortic stent graft, measuring ~ 3.2 x 5.2 x 8.1 cm, increased in size from prior study.

- Diagnostic Procedure(s) and Result(s)
He underwent resection of the lung mass five months after initial presentation and during the surgery, it was noted that the mass was densely adherent to the aortic arch. The lung parenchyma down to the hilum was necrotic with purulent material within it. Pathology demonstrated coalescing necrotizing granulomas with fibrosis and vascular erosions (Figure 4). While bacterial and fungal cultures were negative, AFB smears were positive (Figure 5).
Molecular testing for mycobacterial DNA (GeneXpert MTB-RIF) and 16S rRNA sequencing on the purulent material identified Mycobacterium tuberculosis complex. No rpoB gene mutation was detected. Antimicrobial susceptibility testing showed that the organism was resistant to pyrazinamide but susceptible to rifampin, isoniazid, ethambutol, and streptomycin. Given his history of BCG therapy, the diagnosis of disseminated Mycobacterium bovis- BCG infection was strongly considered, and the isolate was sent for further sequencing to the Centers for Disease control (CDC) reference lab. Sequencing confirmed M. bovis-BCG strain and demonstrated no additional resistance.
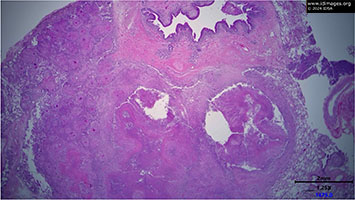 - Figure 4: Pathology; Lung, 1.25x. Coalescing necrotizing granulomas with fibrosis and vascular erosion (Courtesy of Dr. David Sauer).

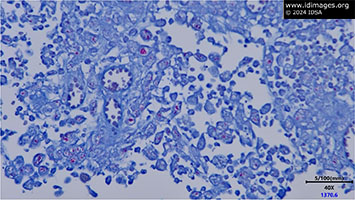 - Figure 5: Pathology Lung, AFB stain, 40x. Multiple aggregates of acid-fast bacilli on AFB stain (Courtesy of Dr. David Sauer).

- Treatment and Followup
Infectious Diseases was formally consulted when the AFB were isolated from the surgical cultures, about five months after initial presentation. The patient was initially placed on empiric therapy for disseminated Mycobacterium tuberculosis with rifampin, isoniazid, pyrazinamide, and ethambutol (RIPE). After the susceptibility testing showed that the isolate was resistant to pyrazinamide, it was stopped. This susceptibility pattern in the setting of previous BCG exposure suggested that this organism was M. bovis- BCG rather than M. tuberculosis.
Treatment was complicated first by rash and oral mucosal ulcers thought to be a drug eruption related to ethambutol. He later developed severe thrombocytopenia requiring steroids and intravenous immunoglobulins (IVIG), ultimately diagnosed as rifampin-induced immune thrombocytopenia (ITP). He was ultimately switched to therapy with isoniazid and levofloxacin for disseminated M. bovis- BCG infection with a plan for a minimum of 12 months of treatment.
The patient presented again to the hospital about 6 months after starting treatment with repeat episodes of hemoptysis. Upper endoscopy was performed, and the findings were consistent with aorto-esophageal fistula, thought to be associated with the disseminated M. bovis BCG infection. The treatment for this would involve esophagectomy along with aortic resection. Due to the high risk of this procedure, the patient declined further treatment. - Discussion
Mycobacterium bovis- BCG is typically a nonpathogenic species of M. bovis which was developed in the 1920s from over 200 re-cultures of M. bovis. It was initially produced as a tuberculosis vaccine and has since also become a standard immunotherapeutic treatment for high-risk non-muscle-invasive bladder cancer [1]. This treatment is typically well tolerated, but local and systemic BCG infections can occur. The incidence of BCG infection following bladder instillation varies from 1%-4.3% in different studies. About half (34.4%-56.1%) of these cases involve disseminated infections [2-4]. The BCG organism can remain in the urinary tract for over a year and disruption of the urothelial barrier from instillation soon after transurethral resection of the bladder, traumatic catheterization, or urinary tract infection may lead to hematogenous spread of the organism [4]. This patient underwent TURBT prior to BCG instillation, which is a known predisposing factor for disseminated infection.
M. bovis is part of Mycobacterium tuberculosis complex, which is a group of closely genetically related species including M. tuberculosis, M. bovis (including M. bovis- BCG), M. africanum, M. microti, and M. caprae. These species can all cause disease in humans but differ in epidemiology and drug resistance [5]. Initial molecular testing in this patient’s case identified M tuberculosis complex, and did not discriminate between species within the complex. While infection caused by M. bovis and M. tuberculosis can present similarly, M. bovis is more likely to cause extrapulmonary disease. Due to this patient’s travel history, M. tuberculosis could not be completely ruled out, and therefore therapy with empiric treatment with RIPE was started. M. bovis is intrinsically resistant to pyrazinamide due to a single point mutation in the pcnA gene from C to G at position 169, which impedes the ability of the enzyme pyrazinamidase to convert pyrazinamide into its active form, pyrazinoic acid [6]. Therefore, the empiric regimen for M. bovis excludes pyrazinamide from the standard RIPE therapy for M. tuberculosis. Evidence supporting treatment regimens for M. bovis is overall limited, especially in instances of drug resistance or intolerance.
In this patient with toxicity from rifampin, one of the treatment cornerstones, we must rely on data for multi-drug resistant M. tuberculosis treatment, expert consultation, and individual sensitivities [9,10]. Fluoroquinolones can be used for rifampin resistant M. tuberculosis with demonstrated sensitivity to fluoroquinolones, but data for the efficacy of M. bovis treatment is lacking [11]. This case demonstrates a relatively rare complication of BCG instillation for the treatment of bladder cancer and highlights the paucity of evidence to inform treatment of M. bovis (including M. bovis - BCG), especially if complicated by drug resistance or intolerance.
- Final Diagnosis
- Disseminated Mycobacterium bovis- BCG infection
- References
-
- Guallar-Garrido S, Julián E. Bacillus Calmette-Guérin (BCG) Therapy for Bladder Cancer: An Update. Immunotargets Ther. 2020 Feb 13;9:1-11. doi: 10.2147/ITT.S202006. PMID: 32104666; PMCID: PMC7025668.
PMID:32104666 (PubMed abstract)
- Nurminen P, Ettala O, Uusitalo-Seppälä R, Nummi A, Järvinen R, Antti K, Boström PJ. Incidence of and mortality from Bacille Calmette-Guérin (BCG) infections after BCG instillation therapy. BJU Int. 2022 Jun;129(6):737-743. doi: 10.1111/bju.15608. Epub 2021 Oct 25. PMID: 34617382.
PMID:34617382 (PubMed abstract)
- Larsen ES, Nordholm AC, Lillebaek T, Holden IK, Johansen IS. The epidemiology of bacille Calmette-Guérin infections after bladder instillation from 2002 through 2017: a nationwide retrospective cohort study. BJU Int. 2019 Dec;124(6):910-916. doi: 10.1111/bju.14793. Epub 2019 May 31. PMID: 31054198.
PMID:31054198 (PubMed abstract)
- Pérez-Jacoiste Asín MA, Fernández-Ruiz M, López-Medrano F, Lumbreras C, Tejido Á, San Juan R, Arrebola-Pajares A, Lizasoain M, Prieto S, Aguado JM. Bacillus Calmette-Guérin (BCG) infection following intravesical BCG administration as adjunctive therapy for bladder cancer: incidence, risk factors, and outcome in a single-institution series and review of the literature. Medicine (Baltimore). 2014 Oct;93(17):236-254. doi: 10.1097/MD.0000000000000119. PMID: 25398060; PMCID: PMC4602419.
PMID:25398060 (PubMed abstract)
- Bayraktar B, Bulut E, Baris AB, Toksoy B, Dalgic N, Celikkan C, Sevgi D. Species distribution of the Mycobacterium tuberculosis complex in clinical isolates from 2007 to 2010 in Turkey: a prospective study. J Clin Microbiol. 2011 Nov;49(11):3837-41. doi: 10.1128/JCM.01172-11. Epub 2011 Sep 21. PMID: 21940474; PMCID: PMC3209075.
PMID:21940474 (PubMed abstract)
- Hannan MM, Desmond EP, Morlock GP, Mazurek GH, Crawford JT. Pyrazinamide-monoresistant Mycobacterium tuberculosis in the United States. J Clin Microbiol. 2001 Feb;39(2):647-50. doi: 10.1128/JCM.39.2.647-650.2001. PMID: 11158123; PMCID: PMC87792.
PMID:11158123 (PubMed abstract)
- Kaur A, Bhandari RK, Rohilla R, Shafiq N, Prakash G, Mothsara C, Pandey AK, Malhotra S. Anti-tubercular therapy (ATT) induced thrombocytopenia: A systematic review. Indian J Tuberc. 2023 Oct;70(4):489-496. doi: 10.1016/j.ijtb.2023.04.029. Epub 2023 Apr 29. PMID: 37968056.
PMID:37968056 (PubMed abstract)
- George JN, Aster RH. Drug-induced thrombocytopenia: pathogenesis, evaluation, and management. Hematology Am Soc Hematol Educ Program. 2009:153-8. doi: 10.1182/asheducation-2009.1.153. PMID: 20008194; PMCID: PMC4413903.
PMID:20008194 (PubMed abstract)
- Lan Z, Bastos M, Menzies D. Treatment of human disease due to Mycobacterium bovis: a systematic review. Eur Respir J. 2016 Nov;48(5):1500-1503. doi: 10.1183/13993003.00629-2016. Epub 2016 Aug 18. PMID: 27540021.
PMID:27540021 (PubMed abstract)
- Nahid P, Dorman SE, Alipanah N, Barry PM, Brozek JL, Cattamanchi A, Chaisson LH, Chaisson RE, Daley CL, Grzemska M, Higashi JM, Ho CS, Hopewell PC, Keshavjee SA, Lienhardt C, Menzies R, Merrifield C, Narita M, O'Brien R, Peloquin CA, Raftery A, Saukkonen J, Schaaf HS, Sotgiu G, Starke JR, Migliori GB, Vernon A. Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines: Treatment of Drug-Susceptible Tuberculosis. Clin Infect Dis. 2016 Oct 1;63(7):e147-e195. doi: 10.1093/cid/ciw376. Epub 2016 Aug 10. PMID: 27516382; PMCID: PMC6590850.
PMID:27516382 (PubMed abstract)
- Mirzayev F, Viney K, Linh NN, Gonzalez-Angulo L, Gegia M, Jaramillo E, Zignol M, Kasaeva T. World Health Organization recommendations on the treatment of drug-resistant tuberculosis, 2020 update. Eur Respir J. 2021 Jun 4;57(6):2003300. doi: 10.1183/13993003.03300-2020. PMID: 33243847; PMCID: PMC8176349.
PMID:33243847 (PubMed abstract)
- Notes
ID Week 2024 - Fellows' Day
Apoorva Bhaskara, MD - Oregon Health and Science University
Contributors: Dr. Radhika Sheth, Dr. David Sauer, and Dr. Christopher Pfeiffer
- Citation
- If you refer to this case in a publication, presentation, or teaching resource, we recommend you use the following citation, in addition to citing all specific contributors noted in the case:
Case #24007: A case of a man with hemoptysis and weight loss [Internet]. Partners Infectious Disease Images. Available from: http://www.idimages.org/idreview/case/caseid=618
- Other Resources
-
Healthcare professionals are advised to seek other sources of medical information in addition to this site when making individual patient care decisions, as this site is unable to provide information which can fully address the medical issues of all individuals.
|
|